Over recent years, numerous companies have achieved significant milestones, and one standout example is Catheter Precision Inc (NYSE American: VTAK). The company has experienced notable growth, particularly with its flagship product, Catheter Precision's VIVO™ (View Into Ventricular Onset). VIVO™ is a non-invasive 3D imaging system designed to help physicians pinpoint the origin of ventricular arrhythmias before procedures, thus enhancing workflow efficiency and reducing procedure duration. It has received marketing clearance from the U.S. FDA and is commercially available in the EU.
HC21 (Healthcare 21), the leading independent medical device distributor in Europe, serving as the United Kingdom distributor, began distributingand integrated VIVO™ in September 2021.Since then, 165 procedures have been performed using VIVO™, averaging approximately 9 procedures per month. With each procedure yielding $1500 from disposables for Catheter Precision,at a gross margin of 90%.Catheter Precision's VIVO™ employs a razor blade model akin to razors and blades, focusing on device placement and revenue generation from disposables.
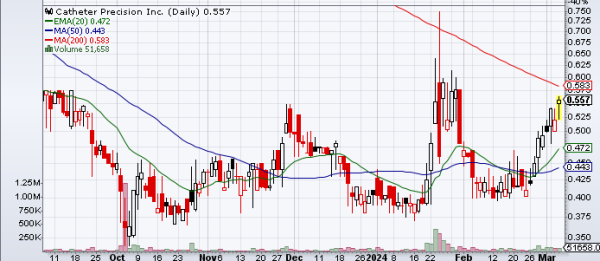
Catheter Precision, Inc. (NYSE American: VTAK) is nearing its 200-day moving average (DMA) and a breakout point at $0.59, maintaining a steady trading pattern above both its 20 and 50 DMA's. The convergence of numerous bullish indicators, coupled with a robust upward trend, suggests that breaching the $0.59 threshold could propel the stock into a higher trading range of $0.75 to $1. Currently in a consolidation phase, the stock shows signs of poised movement.
Established in 2003, HC21 has emerged as one of the rapidly expanding sales, marketing, and distribution entities in the MedTech sector. With annual revenues exceeding 180 million euros, the company employs over 500 individuals across the United Kingdom and Ireland. Collaborating with top-tier medical device firms, HC21 assists them in swiftly attaining their sales objectives.
Catheter Precision's VIVO™ product is a non-invasive localization device designed to detect ventricular arrhythmias before heart procedures. The system comprises hardware, available for one-time purchase, along with a disposable component for single use. The VIVO™ system's software comes pre-installed on a laptop and includes a 3D camera and additional accessories.Currently, Catheter Precision additional market segments, including products such as LockeT, a suture retention device designed to aid in hemostasisfollowing percutaneous venous punctures. LockeT is a FDA-registered Class 1 device. Additionally, the company offers Amigo, a Remote Catheter System, which features a robotic arm for catheter control.
Disclaimers: The Private Securities Litigation Reform Act of 1995 provides investors with a safe harbor with regard to forward-looking statements. Any statements that express or involve discussions with respect to predictions, expectations, beliefs, plans, projections, assumptions, objectives, goals, and assumptions about future events or performance are not statements of historical fact and may be forward looking statements. Forward looking statements are based on expectations, estimates, and projections at the time the statements are made that involve a number of risks and uncertainties that could cause actual results or events to differ materially from those presently anticipated. Forward looking statements in this action may be identified through use of words such as projects, foresee, expects, will, anticipates, estimates, believes, understands, or that by statements, indicating certain actions & quotes; may, could or might occur Understand there is no guarantee past performance is indicative of future results. Investing in micro-cap or growth securities is highly speculative and carries an extremely high degree of risk. It is possible that an investor's investment may be lost or due to the speculative nature of the companies profiled. TheStreetReports (TSR) is responsible for the production and distribution of this content."TSR" is not operated by a licensed broker, a dealer, or a registered investment advisor. It should be expressly understood that under no circumstances does any information published herein represent a recommendation to buy or sell a security. "TSR" authors, contributors, or its agents, may be compensated for preparing research, video graphics, podcasts and editorial content. "TSR" has not been compensated to produce content related to "Any Companies" appearing herein. As part of that content, readers, subscribers, and everyone viewing this content are expected to read the full disclaimer in our website.
Media Contact
Company Name: The Street Reports
Contact Person: Editor
Email: Send Email
Country: United States
Website: http://www.thestreetreports.com






