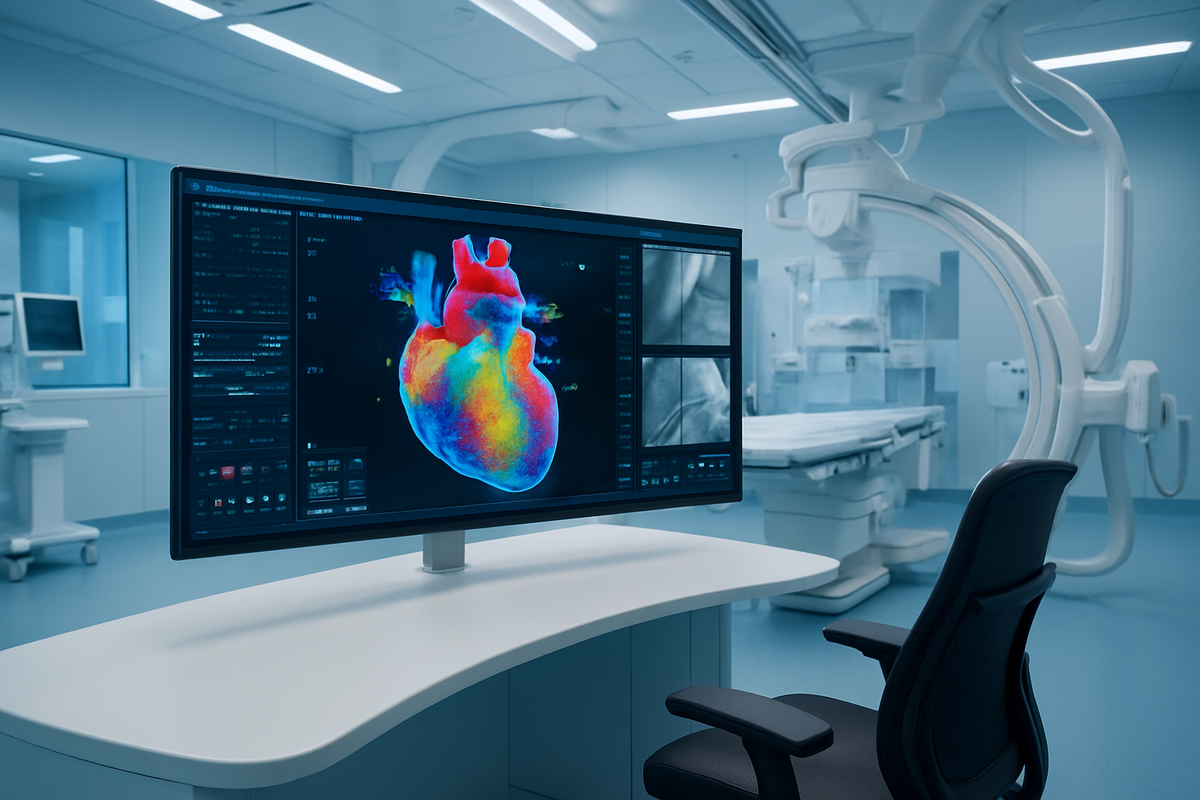
ST. LOUIS – April 6, 2026 – In a milestone that signals a paradigm shift for cardiac care, Stereotaxis (NYSE American: STXS) has received FDA 510(k) clearance for its Synchrony™ System. The approval marks the official commercial launch of the world’s first integrated "digital cockpit" for robotic electrophysiology (EP), a move that industry analysts suggest could finally bring the benefits of robotic precision to the mass market for heart rhythm treatments.
By consolidating disparate diagnostic and therapeutic technologies into a single, unified interface, the Synchrony System promises to solve the "complexity crisis" currently facing cardiac catheterization labs. For patients suffering from complex arrhythmias, the clearance means a move toward more consistent, safer, and highly personalized procedures that leverage the sub-millimeter precision of robotic magnetic navigation.
Bridging the Gap: The Launch of a Digital Ecosystem
The FDA clearance of the Synchrony System is the culmination of a multi-year transformation for Stereotaxis (NYSE American: STXS). While the company has long been known for its pioneering Robotic Magnetic Navigation (RMN) hardware, Synchrony represents its evolution into a software-centric, vertically integrated technology provider. The system centers around a 55-inch, 4K ultra-high-definition display that aggregates real-time data from various sources, including 3D mapping systems, fluoroscopy, and the robotic controls themselves. This allows a physician to perform a complete heart procedure from a seated, ergonomically optimized workstation, rather than standing at the bedside in heavy lead aprons.
The timeline leading to today’s announcement was accelerated by several key regulatory wins over the last six months. In late 2025, Stereotaxis (NYSE American: STXS) secured the CE Mark in Europe, followed closely by the January 2026 FDA approval of the proprietary MAGiC™ Magnetic Interventional Ablation Catheter. The clearance of Synchrony now provides the "connective tissue" required to run these advanced tools. One of the most significant features of the platform is its integration with the EnSite™ X EP System from Abbott (NYSE: ABT), which allows surgeons to view high-density cardiac maps directly within the Synchrony ecosystem.
Market reaction was immediate and overwhelmingly positive. Shares of Stereotaxis (NYSE American: STXS) rose 5.4% in early morning trading as investors processed the news of the commercial launch. Industry stakeholders, including leading electrophysiologists, have praised the system’s "SynX™" cloud integration, which enables real-time remote monitoring and collaboration. This digital-first approach is expected to reduce the steep learning curve traditionally associated with robotic surgery, allowing experts to "proctor" junior colleagues from across the globe in real-time.
The Competitive Shift: Winners and Losers in the EP Market
The most immediate winner of this regulatory milestone is Stereotaxis (NYSE American: STXS) itself. Historically, the company operated on a hardware-heavy model, earning only modest royalties on third-party catheters. With the full Synchrony suite and the MAGiC catheter now cleared, the company has transitioned to a high-margin "razor-and-blade" model. Analysts estimate that revenue per procedure will jump from roughly $1,000 to over $5,000, as the company now captures the full value of the disposable catheters used in every case.
Conversely, the clearance puts pressure on incumbents like Johnson & Johnson (NYSE: JNJ), whose Biosense Webster division has long dominated the manual catheter market. While Biosense Webster remains a titan in the industry, the introduction of Stereotaxis’ own proprietary catheters means a direct loss of royalty and sales volume for J&J in robotic labs. Furthermore, as Synchrony positions itself as a "system-agnostic" digital interface, it threatens to commoditize the hardware from other major players by making the Stereotaxis cockpit the primary point of interaction for the physician.
Other major players, including Boston Scientific (NYSE: BSX) and Medtronic (NYSE: MDT), find themselves at a strategic crossroads. Both have invested heavily in Pulsed Field Ablation (PFA), a fast-growing non-thermal energy source for treating atrial fibrillation. However, Stereotaxis (NYSE American: STXS) has already begun integrating PFA technology through partnerships, such as with CardioFocus. By offering the first robotic PFA solution, Stereotaxis is effectively neutralizing the competitive advantage of manual PFA systems from Boston Scientific and Medtronic, arguing that the speed of PFA is best coupled with the precision of a robot.
Beyond the Lab: Industry Trends and Ripple Effects
The clearance of the Synchrony System fits squarely into two of the most significant trends in 2026: the decentralization of healthcare and the rise of AI-driven surgery. As medical procedures increasingly move from large academic hospitals to Ambulatory Surgery Centers (ASCs), the need for smaller, more efficient, and digitized systems has grown. The Synchrony platform, paired with the recently cleared GenesisX robot—which requires no heavy magnetic shielding—allows community hospitals and outpatient centers to offer high-end robotic care without the multimillion-dollar infrastructure costs of the past.
Moreover, the Synchrony System’s data-rich environment provides a fertile ground for Artificial Intelligence. By capturing every movement and signal in a digital format, Stereotaxis (NYSE American: STXS) is building a massive repository of procedural data. This is expected to lead to the next generation of "autonomous navigation" updates, where AI algorithms suggest the most efficient path for a catheter or identify the precise "sweet spot" for ablation. This mirrors trends seen in general surgery with companies like Intuitive Surgical (Nasdaq: ISRG), but applied to the high-speed, high-signal environment of the beating heart.
From a regulatory standpoint, the FDA's clearance of an integrated ecosystem like Synchrony reflects a growing comfort with software-as-a-medical-device (SaMD). The agency’s focus has shifted toward how these platforms ensure cybersecurity and data privacy, particularly with the remote-access capabilities of the SynX cloud. Stereotaxis (NYSE American: STXS) has set a precedent for how specialized medical device firms can maintain an open-architecture approach—working with partners like Abbott (NYSE: ABT)—while still owning the primary digital real estate of the operating room.
The Road Ahead: What to Watch Next
In the short term, the primary challenge for Stereotaxis (NYSE American: STXS) will be the rapid scaling of its commercial team to support the rollout of Synchrony across the United States. While the technology is revolutionary, the sales cycle for hospital capital equipment remains complex. Investors should watch for the "conversion rate" of the existing installed base of Stereotaxis robots to the new Synchrony platform, as well as early adoption rates within the aforementioned ASC market.
Longer term, the strategic focus is likely to shift toward expansion into other vascular fields. The "Synchrony" architecture was designed to be modular; Stereotaxis (NYSE American: STXS) has already hinted at applying this same robotic cockpit to neurovascular procedures, such as stroke intervention. If the company can successfully prove that robotic navigation is superior for the delicate vessels of the brain as it has for the heart, the total addressable market for the Synchrony platform could triple in size by the end of the decade.
The potential for a strategic acquisition also remains a possibility. Given the high-margin disposable revenue and the proprietary digital platform now in place, Stereotaxis (NYSE American: STXS) has become a more attractive target for a diversified healthcare giant looking to leapfrog into the robotic surgery space. However, for now, the company appears focused on its goal of becoming the "operating system" for the modern interventional suite.
Conclusion: A New Standard for Heart Rhythm Treatment
The FDA clearance of the Stereotaxis Synchrony System is more than just a product launch; it is the formalization of a "Digital EP Lab." By integrating mapping, navigation, and cloud-based collaboration into a single 4K cockpit, Stereotaxis (NYSE American: STXS) has addressed the fragmented and physically demanding nature of heart rhythm treatments. For physicians, it offers a future of ergonomic, data-driven precision; for patients, it offers the promise of safer, more effective outcomes in complex cases that were once deemed too difficult for manual intervention.
As the market moves forward, the "Big Four" medical device companies will likely need to accelerate their own robotic and digital integration efforts to keep pace. The traditional "tools-only" approach to electrophysiology is rapidly becoming obsolete, replaced by ecosystems that prioritize software and connectivity. Investors and healthcare providers alike should closely monitor the adoption of the MAGiC catheter and Synchrony platform throughout 2026, as these will be the primary bellwethers for the success of this robotic revolution.
This content is intended for informational purposes only and is not financial advice.






